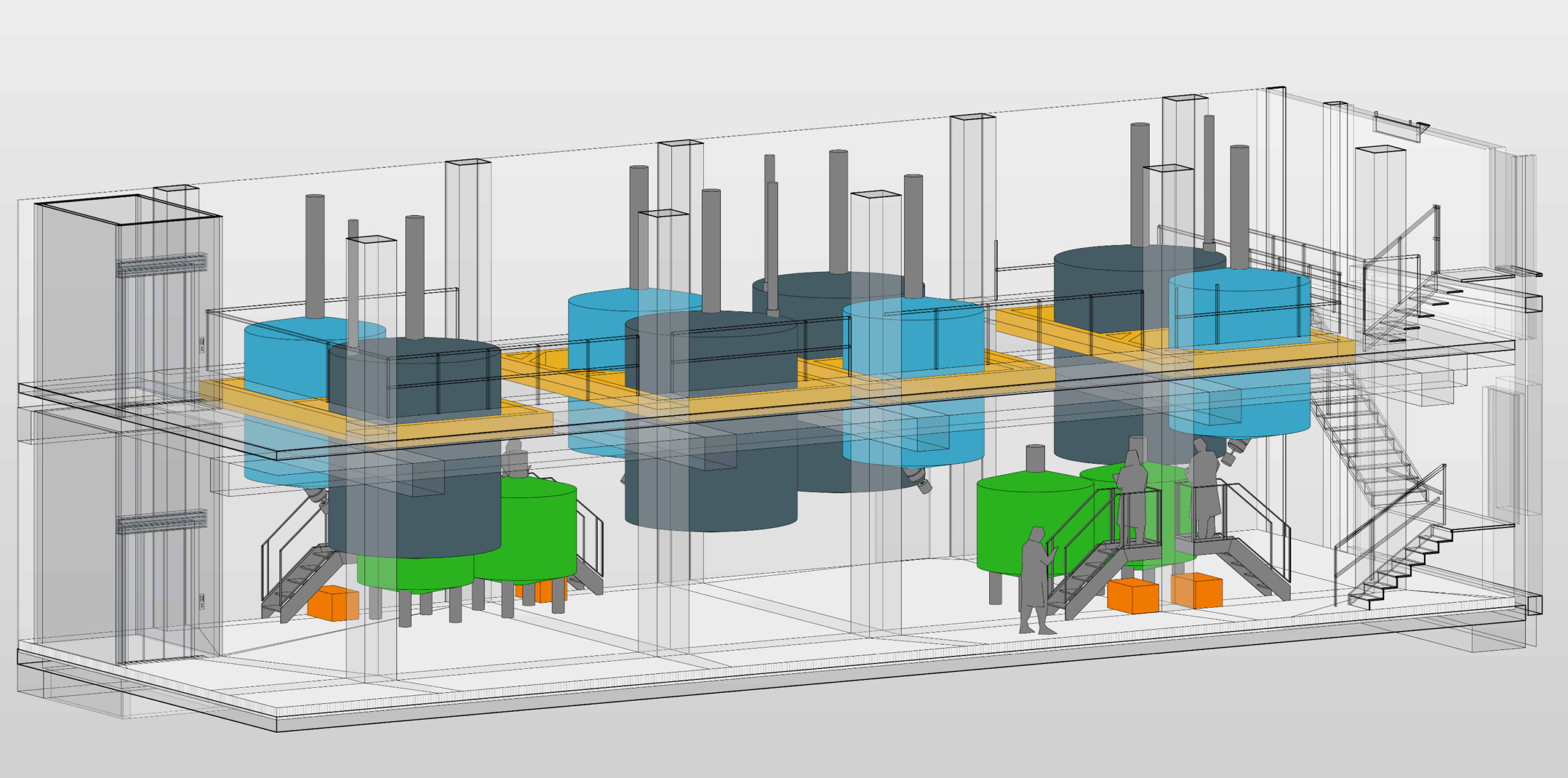
Project
Nordmark is planning to modernize and expand their production for a biological product.
D&N supported Nordmark from an initial feasibility study and the evaluation of design options (greenfield vs. expansion of existing facilities) up to the extended basic design for a brownfield expansion.
Key challenges arose from executing the project during ongoing operations, connecting a new building to existing facilities with outdated structural conditions, and working within very limited construction space. In addition, the planned capacity increase required an evaluation of the site infrastructure, including expansion and modification measures.